Managing Canada’s dual crises
David Renwick of Emergent BioSolutions discusses the challenges of raising awareness for the opioid crisis in the middle of a global pandemic (600 words, 3.5 minutes)
NPC Healthbiz Weekly is presented to you in cooperation with Peak Pharma Solutions
David Renwick and his team at Emergent BioSolutions thought they were on track to addressing many of the challenges inherent in fighting Canada’s opioid crisis when Covid-19 added another hurdle to the mix last year.
Emergent is the biopharmaceutical company behind naloxone (Narcan), a treatment for a temporary reversal of an opioid overdose's effects.
Before Covid-19, there was a growing response to the opioid crisis in Canada from both provincial and federal governments, according to Renwick, the vice president and general manager of Emergent BioSolutions Canada. That progress in the opioid crisis came to an abrupt halt last spring due to the global pandemic.
“The numbers are a little bit disturbing,” said Renwick (photo below) during his presentation at the 14th National Pharmaceutical Congress. “Regionally, the numbers kind of breakdown in an alarming way, there are certain areas that are very concentrated with opioid deaths, and the rates are disturbing.”
According to a CTV News report, in B.C., a total of 1,716 people died due to illicit drug use in 2020, an average of 4.7 deaths per day—a 74 per cent increase in deaths since 2019.
Last month, Toronto Public Health announced the city set a new single-month record after 38 people died due to a suspected opioid overdose.
The dual problems of Covid-19 and the opioid crisis meant Renwick and his team had to re-think their plans for addressing the opioid issue.
“The pendulum swung rapidly in favour of Covid-19 and what that left us with was a government response that was almost unilaterally focused on Covid-19,” Renwick explained. “We took our eye off the ball and what we saw was a resulting impact that was quite significant.”
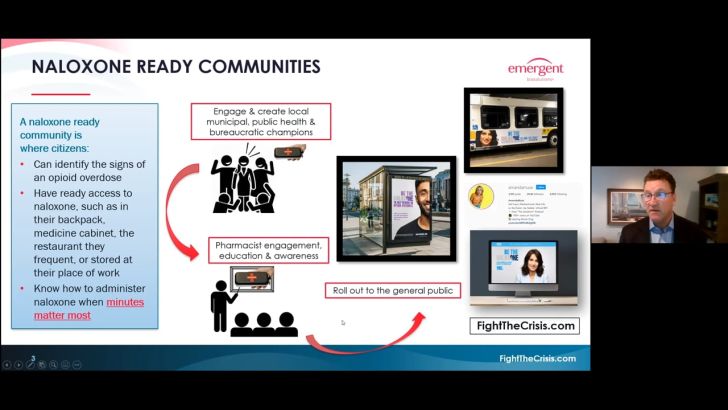
In an attempt to return the attention to the opioid crisis, Emergent developed the naloxone-ready community initiative.
“Minutes matter [when people overdose], and as a consequence of that, we need to get that message out there because it is a life and death situation,” Renwick said. “We wanted to engage and create a kind of local influence.
“We knew that if we had the bureaucratic champions, as well as other stakeholders—be it through public health or pharmacy and others—that we would be well on our way to achieving our goals of creating and building the awareness around [naloxone].”
Since pharmacies stock naloxone, Renwick and his team set out to ensure pharmacists were aware and educated on the benefits of the treatment for opioid overdoses. He noted that despite the media attention and awareness around the opioid crisis, the awareness regarding naloxone remained low at the pharmacy level.
Using traditional print advertising in addition to working with websites and social influencers, Emergent aimed to roll out messaging regarding the importance of naloxone and the naloxone-ready community initiative to the general public.
“The response early on has been overwhelming and positive,” he said. “We are excited to see what the future holds because we believe that this is going to have a tremendous effect on the life and death situation related to opioids.”
The takeaway: Through the dual crisis, Renwick said he learned the importance of communication across various stakeholders.
“Sticking to our message and sticking to the vision of why we are doing this [is key],” he explained. “We needed to make sure that people found a way to understand that they can [handle more than one crisis] at once. I think we have been quite successful in that regard.”
Additionally, the global pandemic has taught Renwick to be innovative using alternative media. Covid-19 forced Renwick and his team to move away from face-to-face sales to remote sales and move from print media to digital and social media strategies.
Further reading: In December, EClinicalMedicine, an open-access clinical journal published by The Lancet, published an article on applying the lessons learned from the Covid-19 response to Canada’s opioid epidemic. Story here.
YOUR HEALTHBIZ WEEK 02/23/21
Eli Lilly will gain an exclusive worldwide license to Rigel Pharmaceuticals’ autoimmune and inflammatory diseases treatment, including its lead candidate, R552, for potentially up to US$960 million, Reuters reports. Rigel will receive an upfront cash payment of US$125 million, with the potential for an additional up to US$835 million in milestone payments. R552 is part of a class of treatments called RIPK1 inhibitors that target a critical signalling protein regulating inflammation and cell death in tissues. It is a new approach to treat various autoimmune, inflammatory, and neurodegenerative disorders. Under the deal, Lilly will assume the costs of global commercialization for the therapy, R552, and Rigel will have the right to co-sell the treatment in the U.S.
Bristol Myers Squibb Canada and Acceleron Pharma announced that Health Canada approved luspatercept for injection (Reblozyl.) It’s Tx of adult patients with transfusion-dependent anemia requiring at least two red blood cell (RBC) units over eight weeks. The indication is for anemia resulting from low-to intermediate-risk myelodysplastic syndromes (MDS) who have ring sideroblasts and who have failed or are not suitable for erythropoietin-based therapy. MDS is a group of cancers that cause limitations in the bone marrow's ability to produce healthy and mature RBCs and typically occur among older people and in more men than women. Luspatercept helps to regulate late-stage RBC maturation to reduce the need for RBC transfusions potentially.
According to this Reuters news report, Novartis has entered into a grant agreement with the Bill & Melinda Gates Foundation to fund the development of accessible single-use gene therapies for sickle cell disease (SCD). The drugmaker said the project's goal is to address the disparity in access to treatments and prioritize populations and regions that bear the greatest burden of hereditary blood disease.
Barrie, Ont.’s Verify Diagnostics announced that Health Canada has authorized the Assure Tech Ecotest Covid-19 serological rapid test to be used at the point-of-care to screen for SARS-CoV-2 antibodies. The approval builds on the U.S. FDA Emergency Use Authorization granted last year. The Assure Tech Ecotest was the first point-of-care fingerstick blood test to be authorized for Clinical Laboratory Improvement Amendments-waived use in the United States and is the first Canadian test to be authorized as well.
NATIONAL PHARMA CONGRESS SPRING WEBINAR
As announced during the National Pharma Congress Winter Webinar, plans are in the works for a spring webinar scheduled for Wednesday, April 7, 2021. Be sure to subscribe to the NPC HealthBiz Weekly for updates on the winter meeting.
CANADIAN HEALTHCARE MARKETING HALL OF FAME
The Canadian Healthcare Marketing Hall of Fame awards were established in 2002 to honour healthcare marketers who have contributed to our avocation and are an inspiration to others.
More than 100 honourees have been selected during the past 18 years. In the selection committee's view, they stand for a representative cross-section of the qualities that make our business unique and fulfilling. Each week, NPC Healthbiz Weekly will acknowledge one past Hall of Fame Honouree.
2012 Inductee
Doug Grant
Bayer Inc.
Toronto
Editor’s note: Doug joined Kyowa Kirin, a research-based life sciences company, as its Canadian General Manager in January 2021.
Doug Grant has had a lifelong affinity for science. Graduating from York University in Toronto with a Bachelor of Science in organic chemistry, Grant envisioned being a laboratory scientist. His first job was in a laboratory, but he quickly discovered that being hands-on in scientific research was not for him.
“Although I wanted to be in science, researching the bench was not my calling,” notes Grant, Senior Vice President and Head, Corporate Affairs, Bayer Inc., where he is responsible for providing management, leadership and strategic direction for communications, trade relations, contracts, pricing, reimbursement, and federal and provincial government affairs. He also oversees the company’s hematology business unit.
Grant began his pharmaceutical career as a sales representative at Dow Pharmaceuticals in Toronto. “My first seven or eight years was spent carrying the bag, doing sales, and sales training,” recalls Grant. “I was learning the base business.”
Grant marketed smoking cessation products such as Nicoderm®, which at the time was not reimbursed. “It was regarded as a lifestyle drug,” he says. “It was unlike today, where people see the value of it. One thing I learned from that experience is the importance of reimbursement.”
The company evolved to Marion Merrell Dow, and in 1995, Grant, a native of the U.S. who spent some of his formative years in Montreal, left Toronto and moved to Quebec, where he joined Pfizer Canada.
Grant had a mandate at Pfizer to build a government reimbursement group. He built further on his knowledge of reimbursement and pricing of products like Lipitor®, Zithromax®, Norvasc®, and Aricept®.
He took a position at Bristol-Myers Squibb Canada, where he refined his negotiating skills in building contracts, focusing on high-profile products like Plavix® and Avapro®. The job offered him a broader perspective around managed care and U.S. markets. He also worked with the company’s communications team, honing his communication skills.
In 2001, Grant returned to Toronto to join Bayer Inc. There were significant challenges from the start. Grant was tasked with managing the withdrawal of the cholesterol-lowering agent Baycol® from the market.
“It was an eye-opener,” says Grant. “I had the fortune to see many of the company’s operations in action, something you do not get to see on a day-to-day basis.”
A couple of months later, Bayer had to respond to demand for Ciprofloxacin® in the wake of global anthrax scares, and Grant was very involved in the issue. “Cipro was tremendously sought after,” says Grant.
Reflecting on a hectic start at Bayer, Grant notes that some of the biggest lessons come out of tough times.
Grant continues to be an industry builder and is inspired by the value of Bayer’s therapies to patients.
“When you see infant boys [with hemophilia] being infused with recombinant factor VIII to help them live, you see how impactful therapy is,” he says.
NEXT WEEK
The 03/02 edition of NPC Healthbiz Weekly will feature Andrew Casey, President and CEO of BIOTECanada, on the new government-industry relationships which have emerged during Covid-19. It’s easy to get your no-charge subscription and have the issue sent to your phone or inbox each Tuesday at 6:00 a.m. sharp.
Stay safe, stay sure, and stay on your game. We’ll see you again next Tuesday.